Polymers | Free Full-Text | α,ω-Epoxide, Oxetane, and Dithiocarbonate Telechelic Copolyolefins: Access by Ring-Opening Metathesis/Cross-Metathesis Polymerization (ROMP/CM) of Cycloolefins in the Presence of Functional Symmetric Chain-Transfer Agents

Radical Ring-Opening of Oxetanes Enabled by Co-Catalysis | Organic Chemistry | ChemRxiv | Cambridge Open Engage
The I-V characteristics of organic hole-only devices based on crosslinked hole-transport layer | Journal of Applied Research and Technology. JART
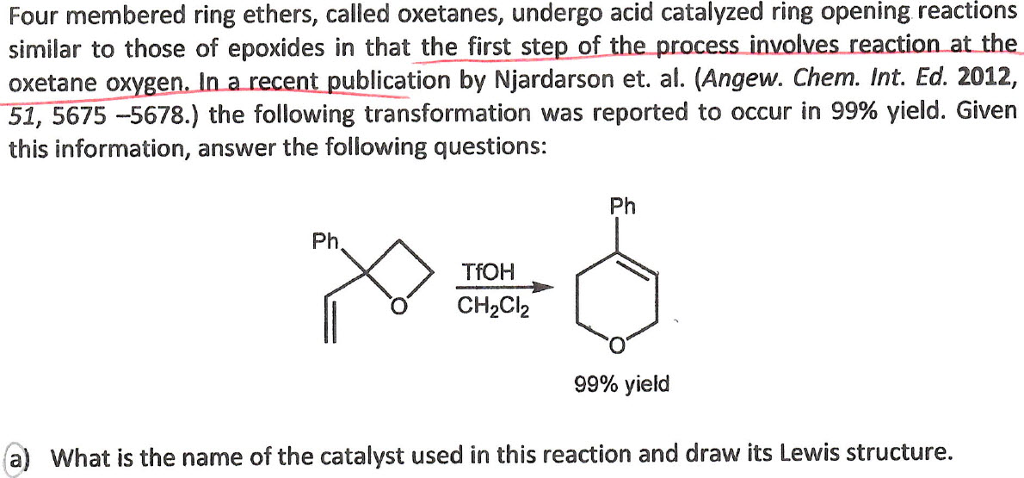
![PDF] Enantioselective Oxetane Ring Opening with Chloride: Unusual Use of Wet Molecular Sieves for the Controlled Release of HCl. | Semantic Scholar PDF] Enantioselective Oxetane Ring Opening with Chloride: Unusual Use of Wet Molecular Sieves for the Controlled Release of HCl. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0efaaee78d740e23578ee345b24b42d34f313217/3-Table4-1.png)
PDF] Enantioselective Oxetane Ring Opening with Chloride: Unusual Use of Wet Molecular Sieves for the Controlled Release of HCl. | Semantic Scholar

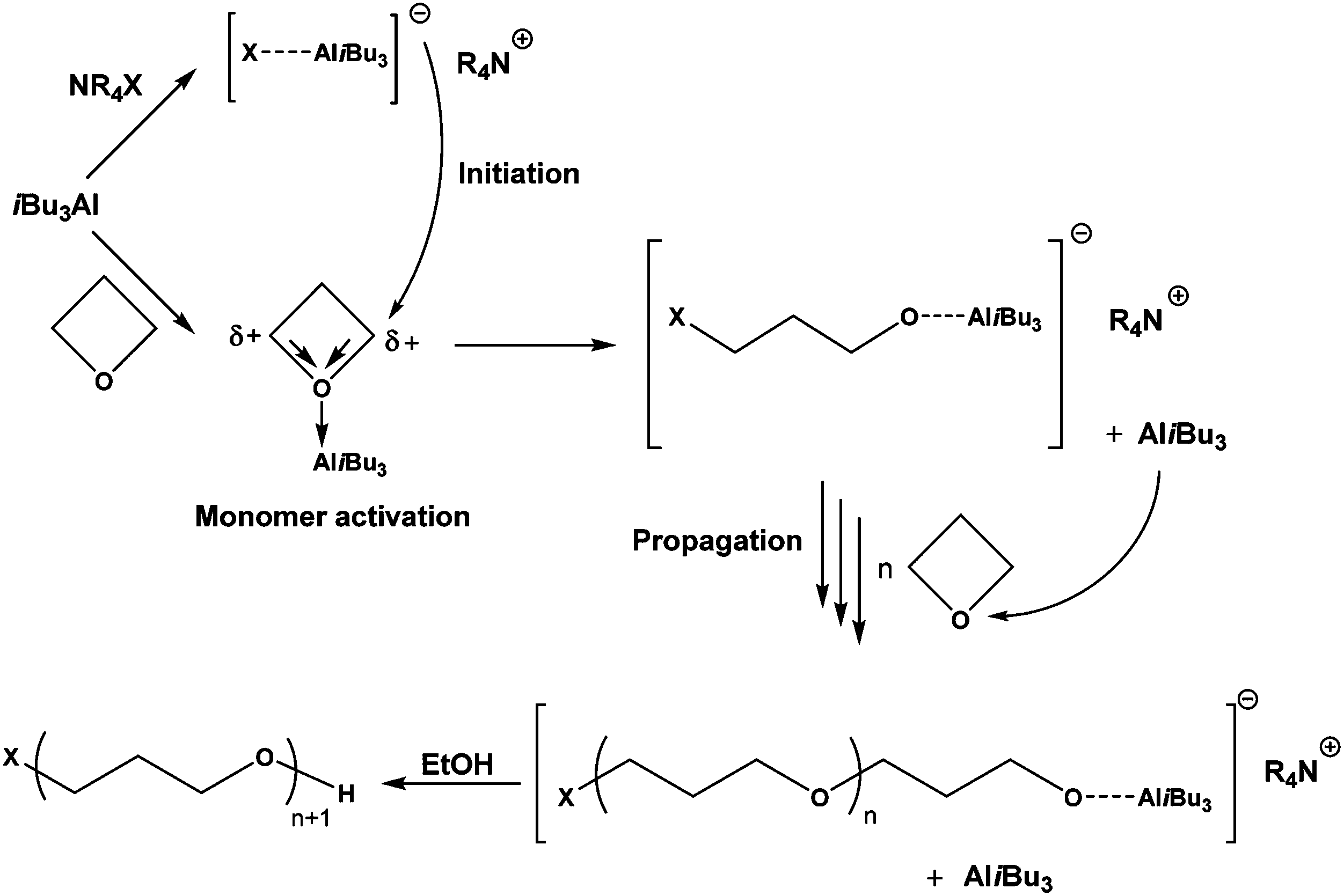
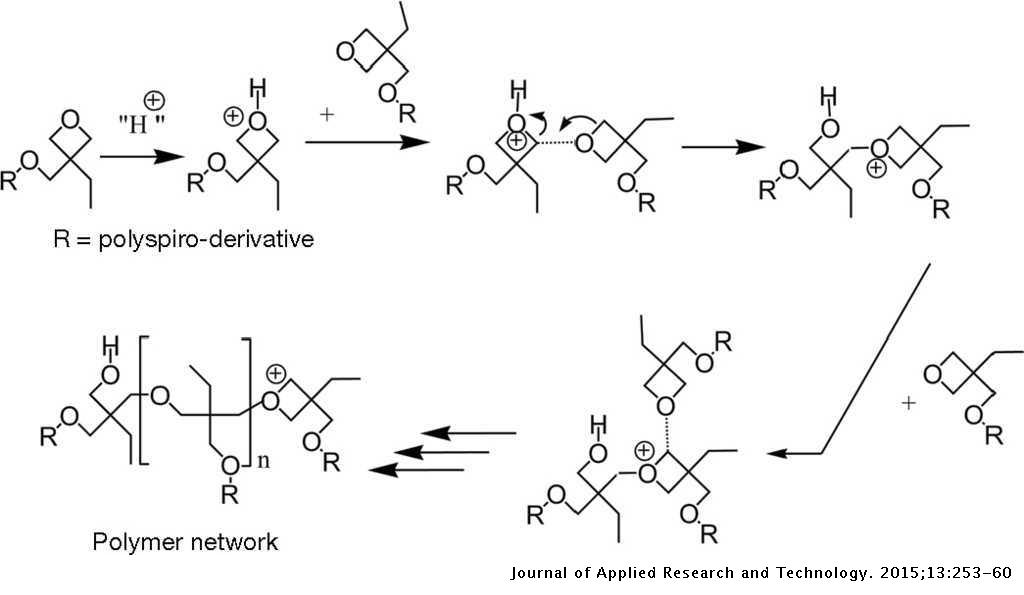
Cationic ring opening polymerization of oxetane proceeds through the... | Download Scientific Diagram
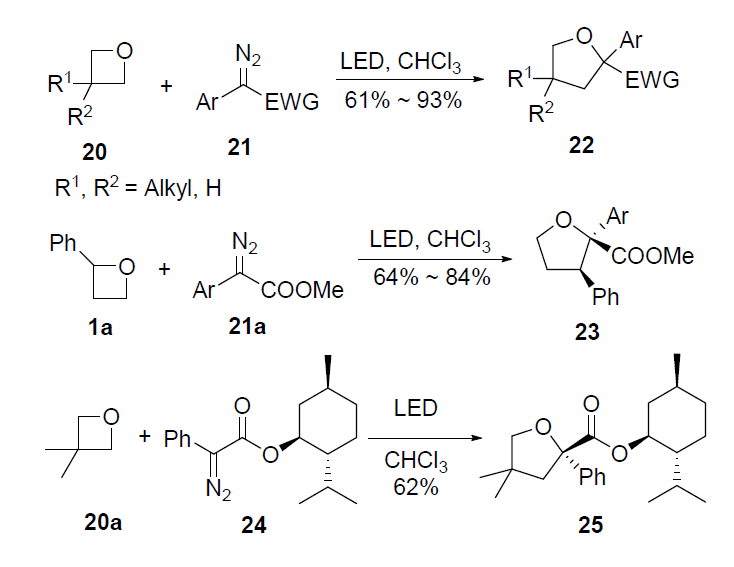
![PDF] A Direct Synthesis of Highly Substituted π-Rich Aromatic Heterocycles from Oxetanes. | Semantic Scholar PDF] A Direct Synthesis of Highly Substituted π-Rich Aromatic Heterocycles from Oxetanes. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d02caafa010dac63526fcccc13673b75609c7a9f/1-Figure1-1.png)
PDF] A Direct Synthesis of Highly Substituted π-Rich Aromatic Heterocycles from Oxetanes. | Semantic Scholar
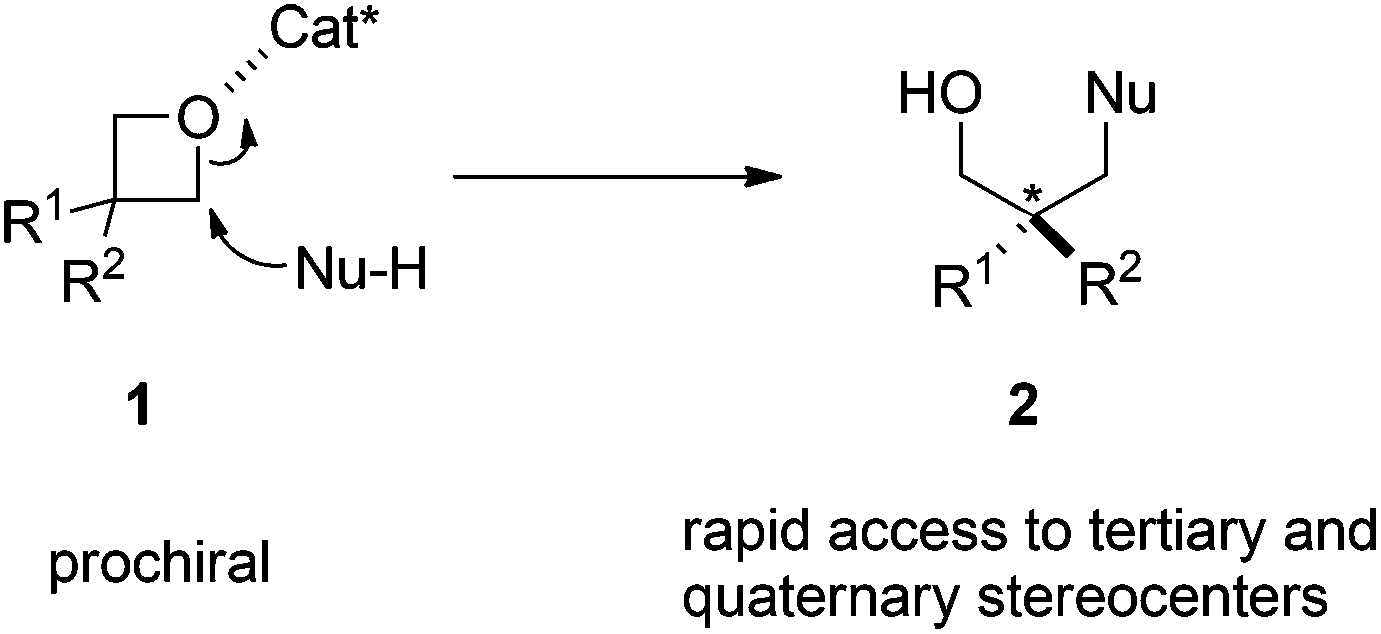
Catalytic asymmetric nucleophilic openings of 3-substituted oxetanes - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB00920G

Cationic ring opening polymerization of oxetane proceeds through the... | Download Scientific Diagram